T023 · What is a kinase?¶
Note: This talktorial is a part of TeachOpenCADD, a platform that aims to teach domain-specific skills and to provide pipeline templates as starting points for research projects.
Authors:
Dominique Sydow, 2021, Volkamer lab, Charité
Talia B. Kimber, 2021, Volkamer lab, Charité
Andrea Volkamer, 2021, Volkamer lab, Charité
Aim of this talktorial¶
In this talktorial, we will talk about kinases: why are they important in life and drug design, what do they look like, and what data resources are available? Finally, we select a set of kinases which will be analyzed in the forthcoming talktorials T024-T028 with respect to their similarity, the goal being to gain insight into potential off-target effects.
Contents in Theory¶
Kinases in a nutshell
The human kinome
Kinase structures and important motifs
Kinase resources
Kinase structures and related information
Bioactivity data
Kinase similarity: Off-targets and promiscuous binding
Kinase dataset compilation
Contents in Practical¶
Define the kinases of interest
References¶
Kinases as drug targets: Nat. Rev. Drug Discov. (2021), 20(7), 551-569
Sequence-based kinase clustering: Manning et al. Science (2002), 298(5600), 1912-1934
KinMap:
Website: http://www.kinhub.org/kinmap/
KLIFS
KLIFS URL: https://klifs.net/
KLIFS database: Nucleic Acid Res. (2020), 49(D1), D562-D569
KLIFS binding site definition: J. Med. Chem. (2014), 57(2), 249-277
Bioactivity data
Karaman et al. dataset: Nature Biotechnology (2008), 26, 127-132
Davis et al. dataset: Nature Biotechnology (2011), 29, 1046-1051
KIBA dataset: J. Chem. Inf. Model. (2014), 54(3), 753-743
PKIS dataset: PLOS ONE (2017), 12, 1-20
Kinase selection: Molecules (2021), 26(3), 629
[1]:
import sys
if "google.colab" in sys.modules:
%pip install teachopencadd --no-deps -q
!teachopencadd -d 23
%pip uninstall teachopencadd -y -q
%pip install -qr requirements.txt
Theory¶
Kinases in a nutshell¶
Kinases are established drug targets to combat cancer and inflammatory diseases (Nat. Rev. Drug Discov. (2021), 20(7), 551-569). They are involved in most aspects of cell life by phosphorylating, and thereby activating, either themselves or other proteins. They are among the most frequently mutated proteins in tumors.
As of September 2021, \(5782\) X-ray structures of human kinases have been resolved (see the KLIFS database) and \(67\) FDA-approved small molecule protein kinase inhibitors are on the market (see the list compiled by the Blue Ridge Institute for Medical Research). Most of the approved drugs bind in the ATP-binding pocket and intermediate surroundings.
Despite of decades of kinase research, there are still many open challenges:
A large fraction of the kinome is un-/underexplored.
Many kinase inhibitors are promiscuous binders causing off-target effects or enabling polypharmacology.
There are occurrences of drug resistance due to mutations.
The human kinome¶
The human kinome consists of over \(500\) protein kinases, but this number may vary depending on the data resource (see overview on Kinodata).
As reported in Science (2002), 298(5600), 1912-1934, Manning et al. clustered the human protein kinases based on their sequence similarity into eight major groups (AGC, CAMK, CK1, CMGC, RGC, STE, TK, TKL) and one “Other” group for unassigned kinases, as well as atypical kinases. The kinase clustering is visualized as the Manning kinome tree. The kinase resource KinMap enables mapping of kinase data onto that tree, e.g. the number of X-ray structures per kinase as shown in Figure 1.
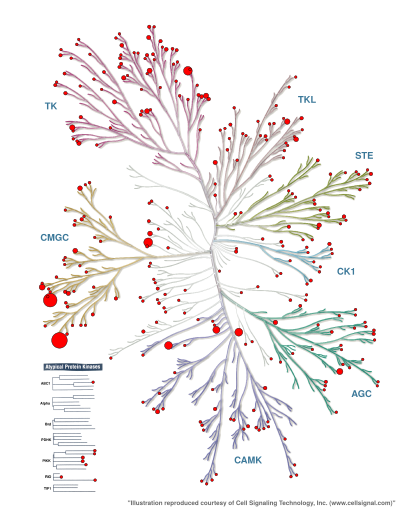
Figure 1: Number of PDB structures per kinase mapped onto the Manning kinome tree using KinMap. Check the appendix on how to generate this KinMap tree.
Kinase structures and important motifs¶
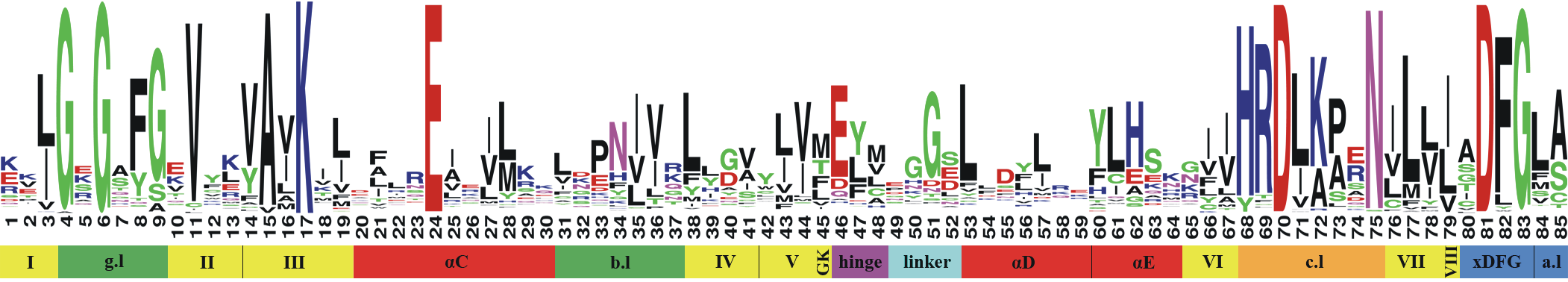
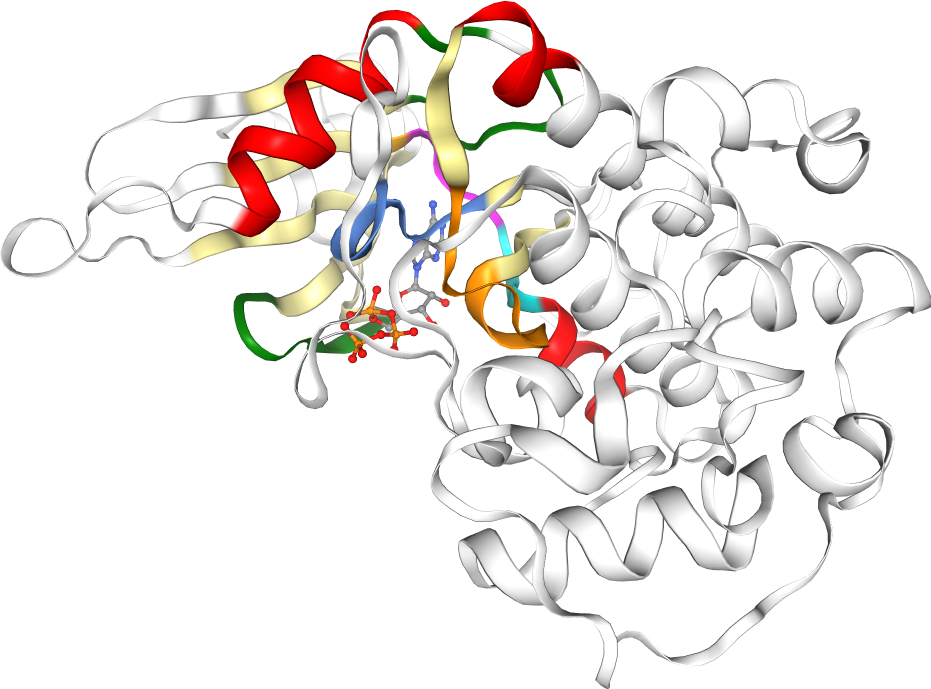
Kinase sequences and structures are highly conserved. Important regions in the kinase pocket include (see Figure 2):
Hinge region: Forms key hydrogen bonds to ligands.
DFG motif: Flips between phenylalanine (F) and aspartate (D), driving the active and inactive state.
αC-helix: Forms in the αC-in conformation a salt bridge between highly conserved lysine (K) and glutamine (Q).
Glycine-rich (G-rich) loop: Stabilizes ATP binding.
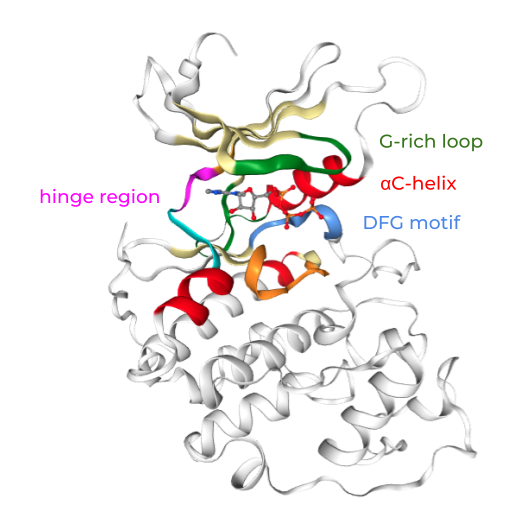
Figure 2: Kinase structure with important key motifs: Hinge region, DFG motif, αC-helix, and G-rich loop. The example structure shown here represents CDK2, PDB ID: 1FIN. Check the appendix on how to generate this visualization with opencadd.
Kinase resources¶
The focus on this protein family has led to a plethora of freely available data on compounds, bioactivity, and structures that are being used for computational drug development (Annual Reports in Medicinal Chemistry 50 (2017): 197-236).
Bioactivity data¶
ChEMBL is a well-known bioactivity database, which releases updated versions every now and then. In September 2021, there are over two million compounds and \(14,000\) targets that are stored. In ChEMBL29, there are over \(160,000\) measurements on kinases (see Figure 4).
kinodataGitHub repository: https://github.com/openkinome/kinodatakinodataChEMBL29 release: https://github.com/openkinome/kinodata/releases/tag/v0.3 (activities-chembl29_v0.3.zip)
As with other data types, the coverage of bioactivity data is highly unbalanced among the human kinases, depending on how much research is spent on certain kinases.
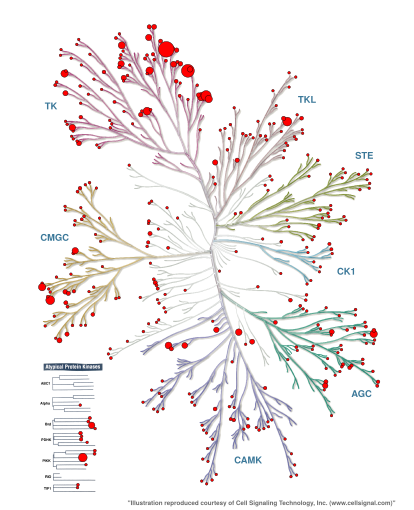
Figure 4: Number of ChEMBL29 bioactivities per kinase mapped onto the Manning kinome tree using KinMap. Check the appendix on how to generate this KinMap tree.
However, ChEMBL is not the only available bioactivity database. Below is an non-exhaustive list of available kinase profiling data sets.
Karaman et al. dataset
Davis et al. dataset
KIBA dataset
PKIS dataset
Paper: PLOS ONE (2017), 12, 1-20
Data: SI data (XLSX)
Kinase similarity: Off-targets and promiscuous binding¶
As described before, kinases are highly conserved, especially in their binding site. This high similarity is a challenge in drug design because ligands may form similar binding modes not only with their designated target (on-target) but also with other targets (off-targets). Such promiscuous binding can cause mild to severe side effects.
Predicting these side effects is non-trivial since some off-targets are not obvious. For example, the EGFR inhibitor Erlotinib shows affinities to other kinases in the highly sequentially-similar TK kinase group. However, it also strongly affects the off-targets GAK, LOK, and SLK, which are in more remote kinase groups (Figure 5).

Figure 5: Profiling data for EGFR inhibitor Erlobinib from the Karaman et al. dataset (Nature Biotechnology (2008), 26, 127-132) mapped onto the Manning kinome tree using KinMap. Check the appendix of this notebook on how to generate this figure.
In the following four talktorials, namely Talktorials T024-027, we will assess kinase similarity from different perspectives, which we compare with each other in Talktorial T028:
Talktorial T024: Kinase similarity based on KLIFS pocket sequence
Talktorial T025: Kinase similarity based on KiSSim pocket structure
Talktorial T026: Kinase similarity based on KLIFS interaction fingerprint
Talktorial T027: Kinase similarity based on ligand promiscuity (ChEMBL bioactivity data)
Talktorial T028: Compare kinase similarity measures from Talktorials T024-T027
Kinase dataset compilation¶
In the course of the kinase similarity talktorials (Talktorials T024-T028), we will use nine kinases from a study published in Molecules (2021), 26(3), 629, which were selected for the following reasons:
Profile 1 combined EGFR and ErbB2 as targets and BRAF as a (general) anti-target.
Out of similar considerations, Profile 2 consisted of EGFR and PI3K as targets and BRAF as anti-target. This profile is expected to be more challenging as PI3K is an atypical kinase and thus less similar to EGFR than for example ErbB2 used in Profile 1.
Profile 3, comprised of EGFR and VEGFR2 as targets and BRAF as anti-target, was contrasted with the hit rate that we found with a standard docking against the single target VEGFR2 (Profile 4).
To broaden the comparison and obtain an estimate for the promiscuity of each compound, the kinases CDK2, LCK, MET and p38α were included in the experimental assay panel and the structure-based bioinformatics comparison as commonly used anti-targets.
Practical¶
[2]:
from pathlib import Path
import pandas as pd
[3]:
HERE = Path(_dh[-1])
DATA = HERE / "data"
Define the kinases of interest¶
We have collected information about these nine kinases in the CSV file T023_what_is_a_kinase/data/kinase_selection.csv:
kinase: Kinase name as used in Molecules (2021), 26(3), 629kinase_klifs: Kinase name as used in the KLIFS databaseuniprot_id: Kinase UniProt IDgroup: Kinase group as defined by Manning et al. Science (2002), 298(5600), 1912-1934full_kinase_name: Full kinase name as used in Molecules (2021), 26(3), 629
[4]:
kinase_selection_df = pd.read_csv(DATA / "kinase_selection.csv")
kinase_selection_df
# NBVAL_CHECK_OUTPUT
[4]:
| kinase | kinase_klifs | uniprot_id | group | full_kinase_name | |
|---|---|---|---|---|---|
| 0 | EGFR | EGFR | P00533 | TK | Epidermal growth factor receptor |
| 1 | ErbB2 | ErbB2 | P04626 | TK | Erythroblastic leukemia viral oncogene homolog 2 |
| 2 | PI3K | p110a | P42336 | Atypical | Phosphatidylinositol-3-kinase |
| 3 | VEGFR2 | KDR | P35968 | TK | Vascular endothelial growth factor receptor 2 |
| 4 | BRAF | BRAF | P15056 | TKL | Rapidly accelerated fibrosarcoma isoform B |
| 5 | CDK2 | CDK2 | P24941 | CMGC | Cyclic-dependent kinase 2 |
| 6 | LCK | LCK | P06239 | TK | Lymphocyte-specific protein tyrosine kinase |
| 7 | MET | MET | P08581 | TK | Mesenchymal-epithelial transition factor |
| 8 | p38a | p38a | Q16539 | CMGC | p38 mitogen activated protein kinase alpha |
We will load this dataset in all downstream talktorials to assess kinase similarity from different perspectives.
Note: You can run the kinase similarity Talktorials T024-T028 with your own set of kinases. To do so, please update the following files:
Update the
T023_what_is_a_kinase/data/kinase_selection.csvfile with your kinases; the only mandatory columns arekinase_klifsanduniprot_id.Update the
T023_what_is_a_kinase/data/pipeline_configs.csvfile with your configurations:Set “DEMO” to 0.
Choose the number of structures per kinases to be used in T025 (KiSSim) and T026 (IFP). If “N_STRUCTURES_PER_KINASE” is set to -1, all structures are used; if set to a number (X), the best X structures are being used for the encoding and comparison (w.r.t. resolution and KLIFS quality score). The latter makes sense for a test run of your data (running the T025 on all structures is time-consuming).
If you run the notebooks on all structures (see “N_STRUCTURES_PER_KINASE”), we recommend to increase the number of cores to be used in T025 (KiSSim) by redefining “N_CORES”.
Let’s take a look at the currently set configurations:
[5]:
pd.options.display.max_colwidth = None
configs = pd.read_csv(DATA / "pipeline_configs.csv")
configs
[5]:
| variable | default_value | description | |
|---|---|---|---|
| 0 | DEMO | 1 | Run the notebooks exactly as displayed online (default: 1) or set to 0 and run your own kinase set (as defined in `kinase_selection.csv`) |
| 1 | N_STRUCTURES_PER_KINASE | -1 | Run structure-based notebooks on all structures per kinase (default: -1) or a subset of structures (replace -1 with e.g. 3) |
| 2 | N_CORES | 1 | Run T025 on one (default: 1) or more cores |
Appendix¶
KinMap data¶
There are some KinMap trees shown in this notebook. The code below generates the KinMap CSV files to be uploaded to KinMap: http://www.kinhub.org/kinmap.
Note:
PNG downloads do not seem to work anymore, thus download as SVG and convert to PNG in your terminal (Linux) via
convert -density 25 my_kinmap_figure.svg my_kinmap_figure.png(SVG cannot be included in Jupyter notebooks out-of-the-box).If SVG download doesn’t render the figure properly, open your favorite text editor and copy paste this into the SVG file:
xmlns:xlink="http://www.w3.org/1999/xlink", resulting in something similar to this in the first few lines:
<svg id="svgCopy" viewBox="0 0 1591 1959" preserveAspectRatio="xMinYMin meet" xmlns="http://www.w3.org/2000/svg" xmlns:xlink="http://www.w3.org/1999/xlink" style=""><desc>Created with Snap</desc><defs></defs><g
[6]:
def format_for_kinmap(kinase_names, kinase_values, size_min=10, size_max=50):
"""
Given kinase names and some associated values, generates a KinMap data file
that will display values as circles of size [`size_min`, `size_max`].
Parameters
----------
kinase_names : list of str
Kinase names.
kinase_values : list of float
Some associated values, such as the number of bioactivites.
size_min : int
Minimum circle size on KinMap tree (minimum input value will be scaled to `size_min`).
size_max : int
Maximum circle size on KinMap tree (maximum input value will be scaled to `size_min`).
Returns
-------
pandas.DataFrame
KinMap data with columns `xName` (kinase name), `size` (circle size for KinMap tree).
"""
data = pd.DataFrame({"xName": kinase_names, "values": kinase_values})
min_ = data["values"].min()
max_ = data["values"].max()
data["size"] = data["values"].apply(
lambda x: ((x - min_) / (max_ - min_) * size_max) + size_min
)
return data[["xName", "size"]]
Number of PDB structures per kinase¶
Generate the number of structures per kinase in the KinMap format to be mapped onto the kinome tree.
[7]:
from opencadd.databases.klifs import setup_remote
klifs = setup_remote()
structures_df = klifs.structures.all_structures()
# Get number of structures per kinase
n_structures_per_kinase = (
structures_df.groupby(["structure.pdb_id", "kinase.klifs_name"])
.first()
.reset_index()
.groupby("kinase.klifs_name")
.size()
)
# Save in KinMap format
kinmap_n_structures_per_kinase = format_for_kinmap(
n_structures_per_kinase.index, n_structures_per_kinase.values
)
kinmap_n_structures_per_kinase.to_csv(DATA / "kinmap_n_structures_per_kinase.csv", index=None)
# Some kinases will not be resolved in KinMap and will be simply dropped
Identify the kinase which has the most structures.
[8]:
kinmap_n_structures_per_kinase.iloc[n_structures_per_kinase.argmax()].xName, max(
n_structures_per_kinase
)
[8]:
('CDK2', 457)
Number of ChEMBL bioactivities per kinase¶
Generate the number of ChEMBL bioactivities per kinase in the KinMap format to be mapped onto the kinome tree.
Note: The cell below takes a few seconds to execute.
[9]:
from opencadd.databases.klifs import setup_remote
# Get bioactivity data
path = "https://github.com/openkinome/kinodata/releases/download/v0.3/activities-chembl29_v0.3.zip"
data = pd.read_csv(path, index_col=None)
data = data[data["activities.standard_type"] == "pIC50"]
data = data.dropna()
# Get kinase data
klifs = setup_remote()
kinases_df = klifs.kinases.all_kinases()
kinases_df = kinases_df[kinases_df["kinase.uniprot"] != "0"]
# Some UniProt IDs have several names in KLIFS, keep only first
kinases_df = kinases_df.groupby("kinase.uniprot").first()
# Map UniProt ID > kinase KLIFS name
data = pd.merge(data, kinases_df, left_on="UniprotID", right_on="kinase.uniprot", how="left")
# Get number of activities per kinase
n_activities_per_kinase = data.groupby("kinase.klifs_name").size()
# Save in KinMap format
kinmap_n_activities_per_kinase = format_for_kinmap(
n_activities_per_kinase.index, n_activities_per_kinase.values
)
kinmap_n_activities_per_kinase.to_csv(DATA / "kinmap_n_activities_per_kinase.csv", index=None)
# Some kinases will not be resolved in KinMap and will be simply dropped
Erlotinib profiling data from Karaman et al. dataset¶
Select “Data Source”: Profiling
Select “Data type”: Karaman et al., 2018
Select “Karaman et al., 2018”: Erlotinib
Click “Add source”
In settings, select “RoyalBlue” in Fill
Click “Apply”
Click on the speech bubble on the top right of the kinome tree to disable annotations.
Note: the name of the on/off-targets (EGFR, GAK, LOK, SLK) have been added manually.
Kinase structure visualization with opencadd¶
We are using as an example the ATP-bound CDK2 structure with the KLIFS ID 4367.
[10]:
from opencadd.structure.pocket import PocketKlifs, PocketViewer
# Get structure and pocket
pocket = PocketKlifs.from_structure_klifs_id(4367)
# Show pocket
viewer = PocketViewer()
viewer.add_pocket(pocket, ligand_expo_id="ATP", show_pocket_center=False)
viewer.viewer.add_ball_and_stick(selection="ATP")
viewer.viewer
[11]:
viewer.viewer.render_image(trim=True, factor=2);
[13]:
viewer.viewer._display_image()
[13]:
[ ]: